- Need immediate help?
- 1 (800)-631-5656
- help@nationalinjuryadvocates.com
Dupixent Lawsuit | Side Effects T-cell lymphoma

Dr. Barry Brock OBGYN: Understanding the 35 Abuse Lawsuits
December 20, 2024
Syfovre Lawsuit | Eye Infections Vision Loss Injury
January 8, 2025Dupixent® (dupilumab), a medication used for eczema, shows potential as an effective treatment for severe asthma. However, researchers caution that it may lead to side effects, including an increase in white blood cells, which could indicate an allergic reaction to the drug.
“Anyone who took Dupixent deserves to know the full picture. If you’ve suffered serious illness without being properly warned about potential risks like CTCL, you deserve justice — and we will fight to ensure your voice is heard and your harms are compensated.” By Attorney Martin Schmidt
The defective drug lawyers at the Schmidt National Law Group is now investigating this drug and it’s harmful side effects, and is currently accepting injury lawsuit cases. If you or someone you know has been harmed by taking Dupixent® and diagnosed with T-cell lymphoma (a type of non-Hodgkin lymphoma that can develop in the blood, lymph system, or internal organs), contact our firm today for a free case review. We expect large cash settlements from the lawsuit process. Call direct at 1-800-631-5656 or use the Secure intake form on this page.
An international research team published their findings in the New England Journal of Medicine back on May 21, 2018 focusing on the safety and efficacy of Dupixent® (dupilumab) for patients with glucocorticoid-dependent severe asthma. The study revealed that Dupixent reduced patients’ reliance on oral glucocorticoids but was associated with a heightened risk of transient blood eosinophilia, characterized by an increase in white blood cells.
Noted Dupixent® side effects include:
- Eosinophilic conditions such as vasculitis (swelling and inflammation of blood vessels)
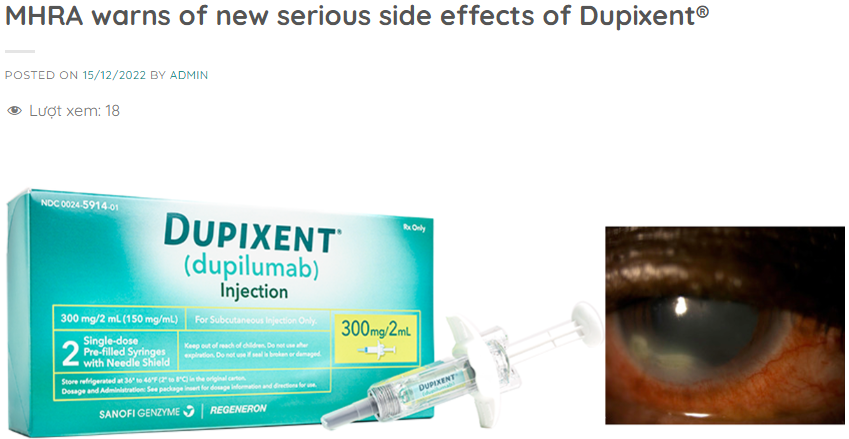
- Eye-related side effects, such as changes in vision and new or worsening eye conditions
Dupixent® Clinical Trial Report
The clinical trial involved 210 patients who were on oral glucocorticoids for severe asthma. Half of the participants received a 300 mg dose of Dupixent® every two weeks for 24 weeks, while the other half received a placebo. Results indicated that Dupixent® decreased the need for steroid treatment and reduced the frequency of severe asthma exacerbations compared to the placebo group.
However, 14% of those treated with Dupixent® experienced transient blood eosinophilia, which is often a sign of an allergic response, and prolonged eosinophilia can lead to tissue damage and other complications. Additionally, the study found that patients receiving Dupixent® experienced more than twice the number of injection-site reactions compared to those on the placebo.
The researchers expressed that the results were expected, as Dupixent® is thought to inhibit the migration of eosinophils into tissues. They noted, “The elevated blood eosinophil counts returned to baseline by the end of the dupilumab treatment period. Patients with temporary increases in blood eosinophil levels did not experience any accompanying clinical adverse events. Since glucocorticoids suppress circulating eosinophils, the more significant reduction in oral glucocorticoid dosage in the dupilumab group compared to the placebo group may have also contributed to the observed eosinophil elevations.”
UK’s MHRA Investigates and Warns on Dupixent®
The following is a direct excerpt from the UK source, link below.
“Up to 7 September 2022, the MHRA has received 479 UK reports which included suspected ocular side effects with dupilumab. 111 of these reports were considered serious.[footnote 2] 9 reports of ulcerative keratitis were received, representing 5 cases (for some individual cases, the MHRA received more than one report from different sources). 2 of these cases involved corneal perforation. 18 reports involved children ranging from 6 to 17 years of age.
With regards to the ocular events listed for dupilumab, the table below summarises the number of UK reports received by the MHRA up to 7 September 2022.[footnote 3]
When a drug is approved and put on the market, patients and their healthcare providers trust the drug is safe.
A few mild side effects might be expected, but some drugs can cause serious complications that were not anticipated by the patient or their doctor. Drug injury lawyers can help those harmed by bad drugs.
The U.S. Food and Drug Administration is responsible for vetting clinical studies performed by drug companies on their products before they are approved. But sometimes those studies are inadequate, resulting in dangerous drugs being sold to consumers.
Patients who were harmed by these dangerous drugs have turned to the lawyers and attorneys at Schmidt National Law Group for over three decades to help then get compensation for their pain and suffering. We have settled thousands of cases for our clients resulting in cash settlements and payouts. Call direct at 1-800-631-5656 or use the Secure Intake Form on this page.
Sources:
https://www.nejm.org/doi/full/10.1056/NEJMoa1804093
https://www.gov.uk/drug-safety-update/dupilumab-dupixentv-risk-of-ocular-adverse-reactions-and-need-for-prompt-management
Dupixent® Class Action Lawsuit page updated on September 19, 2025 to include lawyer claims information.
Learn more about: Can Dupixent Cause Lymphoma?